Calla Health Foundation - Research
Technologies under Calla Health are the Pocket Colposcope and the Callascope, which were developed to implement "see-and-treat" paradigms.
We create innovations that increase wide scale surveillance of cervical pre-cancers and cancers, allowing these countries to tackle their most critical problems—and fight back against preventable disease.
8
Numerous countries have taken part in our clinical investigations and benefited from improved access to women’s healthcare options.
8
We have expanded to 8 sites around the world and continue to grow our reach with powerful women’s health innovations.
1300+
We have enrolled over one thousand patients, providing necessary access to healthcare technology previously outside the reach of these countries.
Backed by:























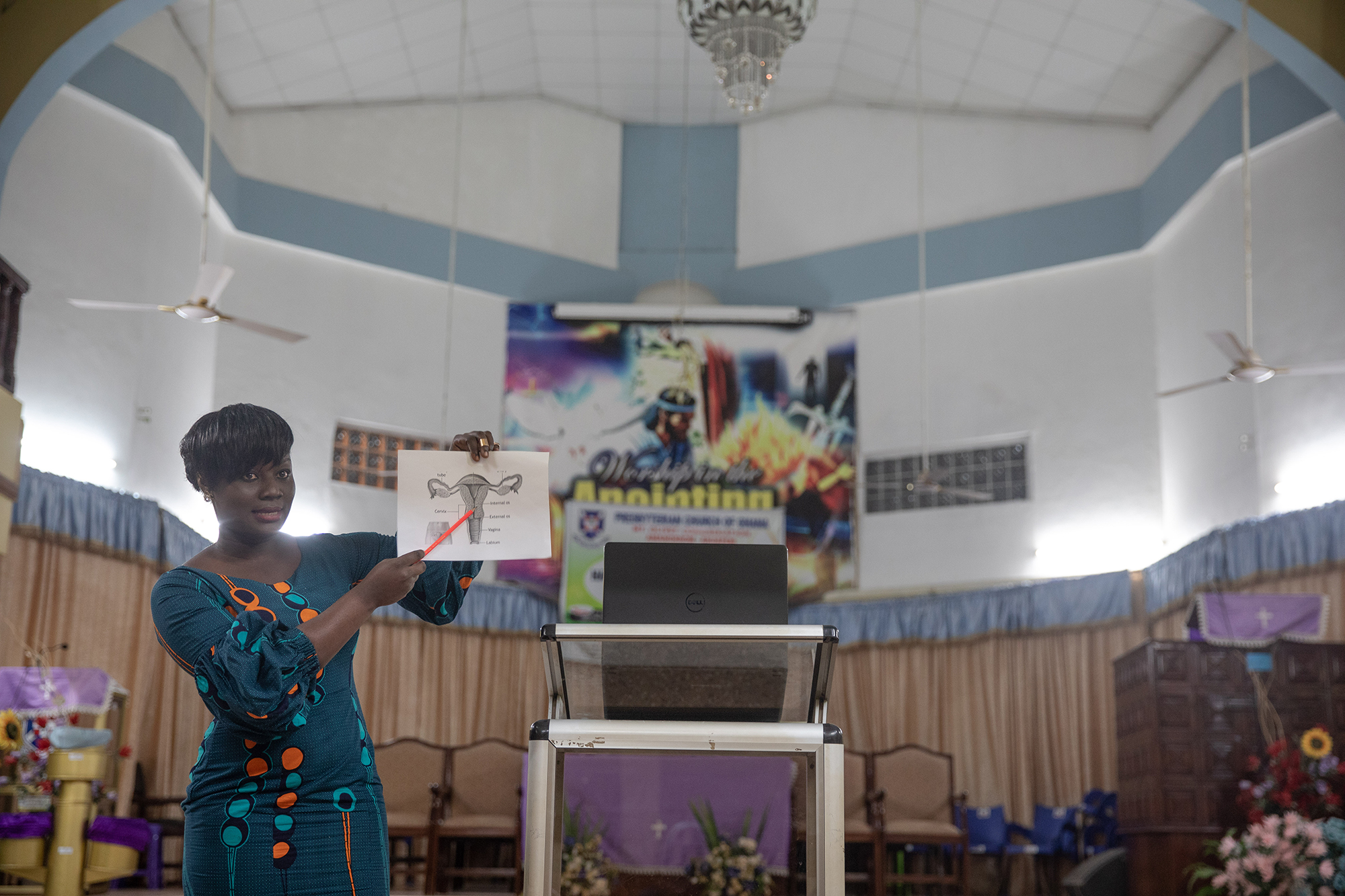
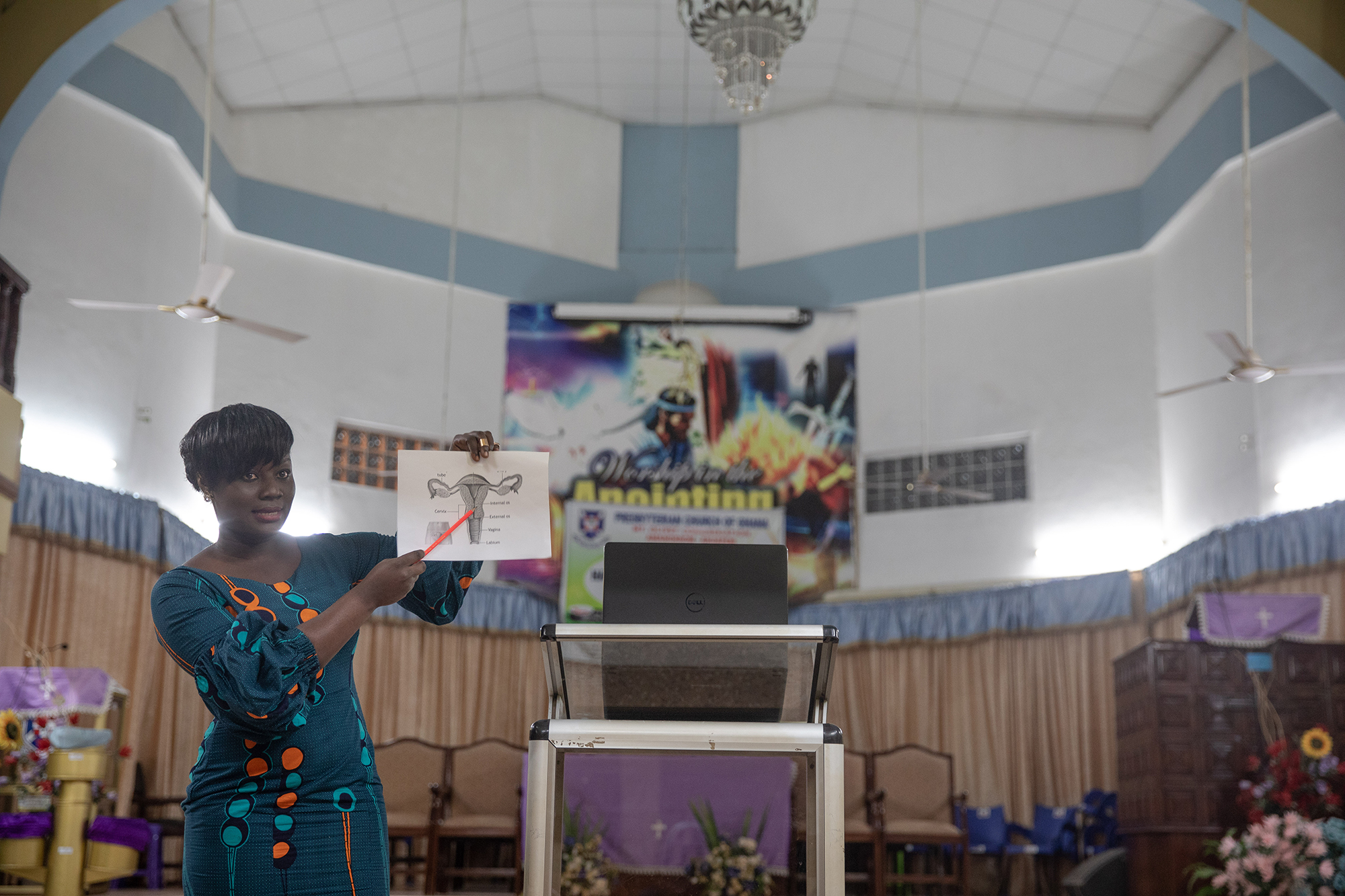


We've conducted clinical investigations in over 8 countries across 4 continents. Our latest product launch, The Pocket Colposcope, received FDA clearance in September 2018.
 Us store
Us store


